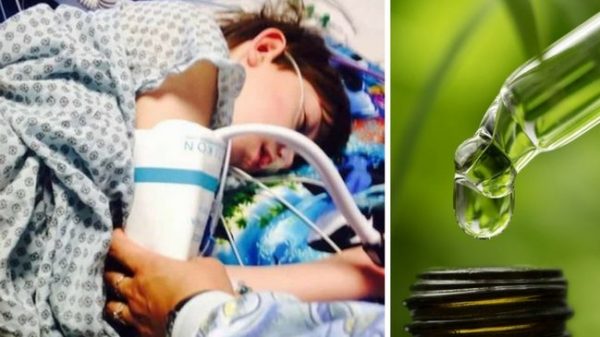
Despite increasing evidence that CBD (cannabidiol), a nonpsychoactive cannabinoid, can help treat severe seizure disorders, a thirteen-year-old Virginia boy suffering from a rare disease has been denied treatment for his use of the plant.
Kaden Hartman suffers from Niemann-Pick disease, also dubbed “Child Alzheimer’s.” According to the National Niemann-Pick Foundation, it is a rare disorder that affects lysosomal storage and metabolism and can cause symptoms including progressive loss of early motor skills, slurred speech, seizures, and hypersensitivity to touch. Most children diagnosed with Niemann-Pick do not live past age 20.
Kaden’s mother, Kathy, contacted High Times magazine to tell his story after he was denied access to an experimental medication he has been taking for three years after the coordinating hospital found out he had also been taking CBD. He has been using the plant-based treatment since he first began experiencing symptoms.
She says Cyclodextrin, the experimental treatment, has been effective in treating his disorder. It was approved directly by the FDA. “When there’s nothing else out there to save his life, the FDA will approve an experimental medicine, and he’s been on it for almost three years,” Kathy told High Times.
She says it’s “definitely working.”
She also believes the CBD is helping reduce his seizures. He has been using it under the supervision of his primary neurologist and initially received a prescription for it from his pediatric neurologist, Dr. Ralph Northam, who is now the governor of Virginia (in Virginia, cannabis oil was approved to treat “intractable epilepsy in 2015, and lawmakers are currently crafting legislation to allow the use of non-psychoactive cannabis oils for a broader range of conditions). This prescription has allowed him to access a more potent version of the drug. Kathy says she told Kaden’s primary neurologist that it “seems to be working great” and says CBD comes with far fewer side effects than traditional anti-seizure medications.
But when Virginia Commonwealth University Medical Center contacted her to tell her Kaden would be taken off Cyclodextrin if he continued to take CBD, Kathy removed CBD from his regimen. “Off CBD, Kaden experiences many seizures. He has since fractured his skull, concussed himself, and developed two blood clots in his brain,” High Times summarized.
“Not only was this 13-year-old denied life-saving treatment because he used CBD oil, but CBD oil itself was a lifesaving treatment for seizures.”
Nevertheless, VCU Medical Center warned Kathy that she had to take her son off the medication. According to her, after they sent the warning letter, they called her, saying “Your son is on CBD oil. He is terminated from the clinical trial. Don’t bother ever showing up again,” she summarized.
She showed up to his next two appointments, anyway, concerned that if she didn’t, the FDA would terminate Kaden’s participation altogether. VCU terminated his spinal tap appointments, which were held every other Monday.
VCU eventually amended their previous statements, saying that “once they have approval from the FDA, the International Review Board (IRB), and the pharmaceutical company,” Kaden could go back on Cyclodextrin.
But, as High Times summarized, “Mrs. Hartman says that they don’t need any of these approvals due to the nature of Kaden’s treatment and that they shouldn’t be sharing information with the pharmaceutical company in the first place.”
She believes doing so could warrant a medical malpractice suit. Worse, getting the approval VCU is demanding could take months. “The FDA only meets once a month. The IRB only meets maybe quarterly or once a month,” Kathy said, worrying that because the company that manufactures the medication has already been bought out twice, Kaden may never be able to go back on it despite the fact that, according to Kathy, he doesn’t even need FDA approval.
“He’s not part of a formal clinical trial. He’s a single patient under the compassionate use law,” she said. Further, she says the trial never forbade CBD. Besides, as Dr. Rebecca Caffrey, a friend of the Hartman family pointed out, “[Kaden] has received many meds not on the protocol, like steroids for the swelling in his fractured skull and pain meds during his recovery from that injury.”
High Times notes that other patients participating in similar drug trials for Niemann-Picks have been able to take CBD with no trouble.
“All they have to do is write a two-paragraph letter to the FDA saying FYI we put him on this medicine,” Kathy says (however, cannabidiol does remain illegal at the federal level, and current compassionate use stipulations direct potential patients to a pharmaceutical version of cannabis). According to her, the doctor in the trial never attempted to do that.
According to 10-WAVY, a regional outlet, the hospital declined to comment on Kaden’s case despite having commented on him previously. The hospital spokesperson cited patient privacy.
In the meantime, Kathy worries that the suspension of Kaden’s trial treatment is endangering his life. She says her friend’s son, who also suffered from the disease, missed two doses of Cyclodextrin and ultimately died. Kaden has missed multiple doses already.
“First do no harm. By doing nothing, you’re creating harm,” as Kathy says she told Dr. Syndi Seinfeld of VCU, who is responsible for Kaden’s treatment.
Or, more bluntly, as she told VCU, “You take him off the study…you just killed my child.”
Source: www.theantimedia.org





That’s not just sick and stupid, it’s depraved.
Thank you. kaden is my son. I went through hell and back because of these idiots